Dry eye disease (DED) encompasses conditions that affect tear production, lid function, and/or epithelial integrity and results in compromise to the ocular surface. Patients who have DED experience symptoms such as irritation, foreign body sensation, itching, and blurry vision of varying intensities.
With severe DED, patients may experience debilitating ocular pain, photophobia, and vision loss. DED is often associated with ocular or systemic inflammatory disease, insults to neural pathways, inherited corneal disorders, and ocular or periocular trauma.
Management algorithms have been recommended for practitioners to follow according to disease stage.1,2 However, this becomes complicated as the disease differs from patient to patient in severity and etiology.3 Many patients who have severe disease fail with supportive therapy alone and require long-term management with consideration of sequelae that may occur.3
This article will review conditions that often present with severe DED—including Sjögren’s, ocular cicatricial pemphigoid (OCP), Stevens-Johnson syndrome (SJS), graft-versus-host disease (GvHD), limbal stem cell deficiency (LSCD), and neurotrophic keratitis (NK)—while highlighting management options beyond traditional supportive and topical ophthalmic therapy.
CONTRIBUTING CONDITIONS
Sjögren’s Sjögren’s is a chronic, multisystem, autoimmune disease mediated by autoantibody production and lymphocytic infiltration that ultimately leads to permanent destruction of the exocrine glands.4,5 Patients who have Sjögren’s often present with ocular irritation, burning, itching, foreign body sensation, blurry or fluctuating vision, in addition to dry mouth.
Clinical findings of Sjögren’s include diminished tear flow, ocular surface staining, increased tear osmolarity, meibomian gland dysfunction (MGD), diminished tear production, filaments, and dilated bulbar conjunctival vessels. Complications of severe ocular Sjögren’s include corneal ulceration and perforation, which may result in corneal melt.4,5
Ocular Cicatricial Pemphigoid OCP is an autoimmune hypersensitivity type II conjunctivitis and is considered a mucous membrane pemphigoid (MMP) subtype. OCP is a chronic relapsing-remitting cicatrizing scarring conjunctivitis characterized by linear deposition of immunoglobulin in the epithelial basement membrane. Recurrent ocular inflammation leads to loss of goblet cells and obstruction of lacrimal gland ducts, resulting in aqueous and mucous tear deficiency.6
Early symptoms include tearing, irritation, burning, and mucus discharge. Chronic conjunctivitis with subepithelial fibrosis will be evident. If left untreated, OCP progresses in up to 75% of patients, resulting in symblepharon formation, entropion, trichiasis, keratopathy, neovascularization, LSCD, severe conjunctival and corneal scarring, ulceration, and keratinization of the ocular surface.7,8 Topical therapy alone is insufficient for these patients and management with systemic immunomodulatory therapies are usually required.7
Stevens-Johnson Syndrome SJS is a rare, acute, severe mucocutaneous reaction, most commonly triggered by viral infections or medications. SJS is considered a delayed hypersensitivity reaction that usually begins with fever and malaise and progresses to exfoliative blisters of the skin involving at least two mucous membranes.9,10
Ocular involvement is asymmetrical and affects up to 50% to 80% of patients who have SJS, ranging from mild dry eye to severe scarring with subsequent ocular surface damage and blindness.9 Secondary to severe inflammation, patients who have SJS may have an inadequate tear film, lid keratinization and trichiasis, LSCD, corneal ulceration, thinning/melting, and perforation.9
Graft-Versus-Host Disease Patients who have a history of leukemia, lymphoma, or multiple myeloma may undergo an allogeneic transplant receiving donor bone marrow or peripheral stem cells as part of their treatment. GvHD is a condition in which donor cells from the graft attack the patient’s (or host’s) organs or tissues.11
Approximately 75% of bone marrow or stem cell transplant recipients are diagnosed with chronic GvHD. Ocular GvHD (oGvHD) is a complication that affects up to 80% of patients who have chronic GvHD. Patients may experience blurry vision, foreign body sensation, burning sensation, severe light sensitivity, chronic conjunctivitis, dry eyes, and eye pain. Ocular involvement includes confluent areas of punctate keratopathy, keratoconjunctivitis sicca, filamentary keratopathy, superior limbic keratoconjunctivitis, symblepharon formation, and cicatricial conjunctivitis.11
Limbal Stem Cell Deficiency Limbal epithelial stem cells reside in specific regions at the limbus known as the limbal stem cell niche. Damage to the stem cells or disruption of the niche may lead to LSCD. In LSCD, the conjunctiva proliferates over the cornea leading to opacification and vascularization, which may result in reduced vision, pain, and photophobia. LSCD has a wide array of etiologies including hereditary or genetic causes, chemical burns, inflammatory ocular surface diseases, contact lens wear, radiation, ocular surgery, topical medications, and others.12
LSCD is often diagnosed based on history and the clinical findings of loss of limbal anatomy, corneal conjunctivalization (whorl-like corneal staining pattern), persistent epithelial defects, and scar formation.12 LSCD diagnosis can be confirmed by impression cytology, and in vivo confocal microscopy and anterior segment optical coherence tomography (AS-OCT) may assist in quantifying the condition.13
Neurotrophic Keratitis NK is a degenerative disease that leads to spontaneous breakdown of corneal epithelium and impaired healing, characterized by a reduction in corneal sensitivity.14 A decrease or loss of corneal sensation is the hallmark finding of NK due to damage to the trigeminal nerve.14 There are numerous causes of NK including herpetic keratitis, ophthalmic and neurosurgical procedures, chemical burns, chronic use of ophthalmic medications, diabetes, congenital syndromes, and intracranial space-occupying lesions.
Mackie developed a three-stage classification of NK based on severity of corneal damage ranging from superficial punctate keratopathy to recurrent and/or persistent epithelial defects (PED) and ulcers, which may further progress to stromal melting and corneal perforation.15,16
MANAGEMENT BASICS
Refractory cases of severe DED can be frustrating for the patient and provider. Ultimately, the goals when managing patients who have severe DED are to promote corneal healing, protect the ocular surface, prevent complications, and restore corneal function. Traditionally, supportive therapy alone was used in the management of these patients.
In recent years, we’ve seen promising novel medical and surgical treatments developed to not only heal but also restore function for patients suffering from severe DED. Treatment should be continued and altered to provide symptom relief, halt disease progression, and protect the ocular surface from environmental or mechanical insult. Management for these patients depends on disease severity and is often chronic. Table 1 lists some possible treatment options.
| • Tear conservation |
| • Environmental management |
| • Eyelid taping or patching |
| • Elimination of offending medications/preservatives |
| • Artificial tears, gels, or ointments |
| • 5% sodium chloride q.i.d. reduces corneal edema |
| • N-acetylcysteine reduces viscosity of mucus in the tear film |
| • Punctal plugs |
| • Topical cyclosporine or lifitegrast |
| • Topical antibiotics |
| • Recombinant human growth factors |
| • Topical steroids |
| • Topical non-steroidal anti-inflammatory drugs (NSAIDs) |
| • Biologics |
| • Amniotic membranes |
| • Moisture glasses |
| • Contact lenses |
Early in the management of DED, all offending topical medications, particularly those containing preservatives such as benzalkonium chloride (BAK), should be eliminated due to their detrimental effects on the ocular surface. Additionally, comorbid ocular and systemic diseases, which may worsen any of the conditions discussed, should be managed aggressively. Management may include any combination of the following: preservative-free artificial tears, gels, and ointments; punctal occlusion; hypertonic drops; N-acetylcysteine; cyclosporine; lifitegrast; antibiotics; corticosteroids; and nonsteroidal anti-inflammatory agents (NSAIDs).1 Use of topical steroids and nonsteroidal anti-inflammatory agents should be used with caution as they inhibit stromal healing and may increase the risk of corneal melting and perforation for some cases.16 Ophthalmic antibiotics are recommended to prevent infection in cases of severe corneal staining and persistent epithelial defects, and topical N-acetylcysteine and systemic tetracycline may be considered to control collagenase activity.17-19 A novel nasal spray, varenicline 0.03mg (recently approved in October 2021), may be beneficial for patients who have compromised ocular surfaces and who cannot tolerate topical medications as it’s applied intranasally.20
BIOLOGICS
These are often used when conventional therapies fail. Biologics are bioengineered molecules used to target cytokines responsible for inflammation and act to suppress the body’s immune system.21 Current available agents available to treat DED are serum tears, amniotic membranes, and cenegermin.
Serum Tears Blood serum shares much of the biologic composition of natural tears and can provide essential epithelial support and antimicrobial activity.22 When the tear film is deficient, serum tears—whether autologous, allogenic, or platelet-rich plasma (PRP)—can play an important role in restoring ocular surface health.
Studies have demonstrated that the use of serum enhances corneal epithelial wound healing due to contained growth factors.23-25 Serum also enhances comfort, improves tear film stability, improves Schirmer scores, inhibits the release of inflammatory cytokines, and increases the number of goblet cells and mucin expression in the conjunctiva.23,24,26
Autologous serum tears (ASTs) These are made from the patient’s own blood sample and are a complex mix of growth factors, proteins, antioxidants, and lipids. Serum is the fluid component of full blood that remains after clotting. After the blood serum is collected, it’s combined with sterile saline to a concentration ranging from 20% to 100% based on the severity of DED and divided into smaller vials. The vials must be kept refrigerated or frozen to avoid contamination. Serum diluted to 20% or less enhances the proliferation of epithelial cells, and 50% or 100% serum stimulates fibroblasts more effectively.1 ASTs can be dosed anywhere from two to eight times per day but are commonly dosed six times per day to q2h for patients who have severe dry eye disease. ASTs are not approved by the U.S. Food and Drug Administration (FDA) or covered by most health insurances.27
Allogenic serum tears These are produced from another patient’s blood and are an equally efficacious alternative for patients who are not suited for ASTs, such as those who have systemic comorbidities or cannot safely partake in frequent blood sampling.28 Allogenic tears may also be an option for patients who have decreased growth factor concentration or high pro-inflammatory mediators in their AS (particularly those who have Sjögren’s and oGvHD), a common finding in patients who have systemic inflammatory disease versus those who have localized DED.28,29 Additional benefits include the ability to produce these in larger quantities, increasing cost effectiveness for the patient. To ensure hemato-immunologic antigen matching, type AB blood donors (the universal plasma donor blood type) are preferred.30
Platelet-Rich Plasma (PRP)/Plasma Rich in Growth Factor (PRGF) To produce PRP/PRGF, collected blood from the patient is centrifuged with an anticoagulant that prevents blood clotting and activation of platelets, thus keeping the platelets in their native form. Centrifugation separates the dense red and white blood cells from the platelets and plasma. The platelets and plasma are collected with a syringe and put in eye drop bottles without being diluted. Because the elimination of platelets in the prep of AST significantly lowers growth factor levels, PRP may hold a therapeutic advantage over the other two types of serum tears. Also, some studies indicate that the AS of patients who have active autoimmune diseases, unlike PRP, have significantly higher amounts of inflammatory cytokines, which are deleterious to the ocular surface and can lead to further damage.31 PRGF eye drops, like PRP drops, have been shown to increase corneal epithelial cell proliferation, accelerate wound healing with reduced scar development, protect against fibrosis, and suppress bacterial growth.32
Serum tears have shown to improve corneal staining and improve persistent epithelial defects in patients who have Sjögren’s, MMP, and GvHD.33 All of the above options can be utilized with and without scleral lenses for patients who have severe DED and PEDs.33 Overall, serum drops promote migration and proliferation of healthy corneal epithelium while lubricating the ocular surface, preventing epithelial adhesion to the tarsal conjunctiva, and reducing shear stress.
Amniotic Membrane Tissue (AMT) This is the innermost, avascular layer of the placenta and aids corneal epithelialization, reduces inflammation and fibrosis, prevents structural damage, and boasts antimicrobial properties.34 It can be used either as a surgical graft or a temporary corneal bandage. It comes as either cryopreserved or dehydrated.
The regenerative properties of AMT have been attributed to a heavy chain hyaluronic acid (HC-HA) bound to pentraxin-3 (PX3), which inhibits pro-inflammatory cells and suppresses T-cells to treat inflammation and prevent scarring.35 Due to higher levels of processing, dehydrated products have the lowest amounts of this molecule, which is the reason the FDA labeled them for wound coverage versus for accelerated wound healing like their cryopreserved counterparts.
For patients who have moderate to severe DED, amniotic membrane therapy can act as a great catalyst to the healing process, enhancing the recovery of the ocular surface and reducing the signs/symptoms of dry eye36 (Figure 1). Studies have shown that cryopreserved AMTs were found to improve symptoms and ocular surface health for an average of more than four months following application with an optimal retention time of five days,37 as well as promote corneal nerve regeneration and improve corneal sensitivity up to three months.38 AMT is a synergistic adjunct therapy to other pharmacologic treatments and should not be a standalone therapy.
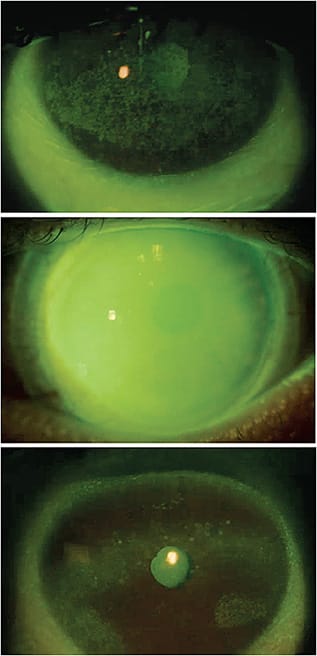
Nerve Growth Factors (NGFs) NGFs stimulate tear secretion, epithelial cell proliferation, and the regeneration and survival of sensory nerves. In clinical studies, 72% of patients treated with an eight-week course of a recombinant NGF achieved complete corneal healing and 80% remained completely healed at one year.39
It is dosed at every two hours (six times per day) for a period of eight weeks. It must be kept refrigerated. It is used to treat nonhealing corneal defects due to stage 2 and 3 NK, although studies are ongoing for the indication of stage 1 NK. A recent study also demonstrated corneal reinnervation and recovery of corneal sensitivity at the end of treatment, as well as evidence that nerve growth and corneal sensitivity continue to increase up to eight months after discontinuation of therapy.40
Intravenous Immunoglobulin Eye Drops (IVIG) IVIG is a new class of biologics being studied in DED related to autoimmune diseases. IVIG are eye drops formulated using pooled antibodies that were created from immune globulins made from the donated blood of thousands of individuals. IVIG contains varied types of antibodies that combat the negative effects of anti-citrullinated protein autoantibodies (ACPAs). ACPAs are a specific type of antibody in the tear film that causes a self-perpetuating cycle of chronic inflammation on the ocular surface in DED and have been found in subtypes, such as Sjögren’s, oGvHD, MGD, and discordant DED (those who have dry eye symptoms but no clinical signs). An ongoing study has found that administering an IVIG drop twice a day for eight weeks caused a significant reduction in the signs and symptoms of DED.41
SCLERAL LENS SOLUTIONS
In addition to new topical therapies and biologics, scleral lenses (SLs) are being prescribed for the management of ocular surface disease (OSD).42,43 When fitting SLs for patients who have OSD, the goal is to mitigate symptoms while protecting and rehabilitating the ocular surface.44 Patients who have severe DED, particularly those with nonhealing PEDs, have found success with SLs, often used in conjunction with other therapies, as their design allows for continuous corneal hydration with no corneal contact.45
The Tear Film & Ocular Surface Society (TFOS) Dry Eye Workshop II (DEWS II) recommended therapeutic contact lenses—both soft bandage contact lenses and rigid scleral lenses—in step 3 of the management and treatment recommendations for DED.1 Patients who have DED are often referred for SLs when other treatments fail. In a survey study, SLs ranked as the fifth choice for ocular surface management after topical lubricants, topical steroids, cyclosporine, and punctal occlusion.43 However, 20% of hospital-based practitioners ranked SLs as their first, second, or third treatment for OSD in the same survey.43
When fitting SLs, the diameter of the SL for patients who have dry eye disease is often dependent on the surface area that needs rehabilitation. The larger the diameter of the lens, the more advanced haptics may be required due to the asymmetry of the sclera. Corneoscleral mapping may be beneficial in these cases to provide further understanding of the ocular surface shape, particularly for those patients who have significant cornea and conjunctival irregularities (Figures 2 and 3). Other patients may benefit from impression-based SL technology, especially as much of our mapping software relies on a decent quality tear film to get a reliable image.
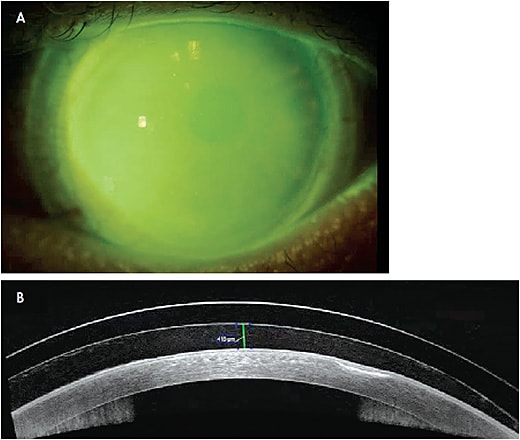
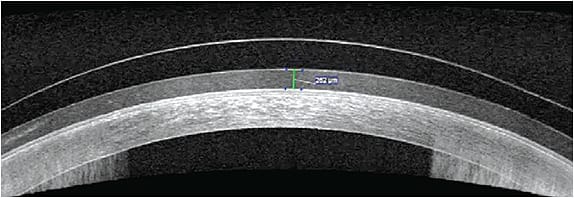
SURGICAL OPTIONS
Patients who have persistent epithelial defects or refractory cases of severe DED, may need surgical intervention including a partial or total tarsorrhaphy, amniotic membrane transplantation, or a conjunctival flap, all aimed at restoring the integrity of the ocular surface.14,46,47 Corneal nerve restoration is now possible with corneal neurotization procedures for patients who have NK.
Tarsorrhaphy During tarsorrhaphy, the superior and inferior eyelids are sewn together to reduce ocular exposure and protect the cornea. This procedure has demonstrated the ability to promote corneal healing across multiple scenarios and is associated with a 90.9% success rate.48 Tarsorrhaphies can be central or peripheral, temporary, or permanent.
Temporary tarsorrhaphies are performed under local anesthesia doing a double pass of nonabsorbable suture through the inferior and superior eyelids. Botox can be injected into the levator to induce a temporary upper eyelid ptosis and can be an alternative to sutures, lasting a mean of 46 days.49 Permanent tarsorrhaphies involve the intramarginal adhesion of the upper and lower eyelids.50 They are utilized when the neurotrophic condition is more severe and maximal therapy has already failed to treat the exposure and the length of time to healing is uncertain. Figure 4 shows an example of using a partial tarsorrhaphy.
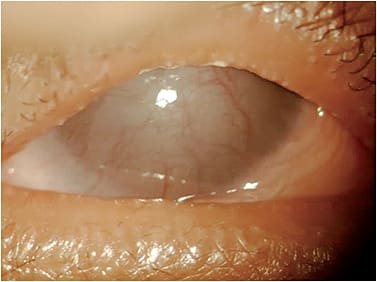
Conjunctival Flaps This involves excising a damaged section of cornea and replacing it with a section of the patient’s own conjunctiva to cover the cornea. The purpose of this surgery is to restore the integrity of the corneal surface and thus prevent gradual corneal ulceration and secondary infection, as well as to ameliorate pain, reduce the need for frequent medications, improve cosmetic appearance, and offer an alternative to invasive surgery or enucleation.51 This procedure is generally underutilized today because of the availability of many alternative therapeutic options.
Corneal Neurotization (CN) CN is a surgical procedure for sensory reinnervation in NK and can significantly improve corneal sensation in all disease stages, no matter the cause, and in some cases, it can also improve visual acuity.52 CN involves transferring healthy donor sensory nerve axons to the denervated cornea to reestablish innervation. On average, there is a maximum return of sensation by eight months.52 It has been demonstrated in both humans and animal models to protect the cornea from ulceration and perforation and to promote faster epithelial healing compared to neurotrophic corneas.53
GOING FORWARD
Advances in technology have significantly improved therapeutic options for patients who have severe DED. In addition to topical ophthalmic management, biologics, scleral lenses, and surgical interventions should be considered for patients unresponsive to traditional therapy. This is an exciting time to be an eyecare practitioner, with ongoing and future developments expected to further enhance our ability to provide patient specific therapies in DED. CLS
REFERENCES
- Jones L, Downie LE, Korb D, et al. TFOS DEWS II management and therapy report. Ocul Surf. 2017 Jul;15:575-628.
- Starr CE, Gupta PK, Faird M, et al. An algorithm for the preoperative diagnosis and treatment of ocular surface disorders. J Cataract Refract Surg. 2019 May; 45:669-684.
- Baudouin C, Aragona P, Van Setten G, et al; ODISSEY European Consensus Group members. Diagnosing the severity of dry eye: a clear and practical algorithm. Br J Ophthalmol. 2014 Sep;98:1168-1176.
- Mariette X, Criswell LA. Primary Sjögren’s Syndrome. N Engl J Med. 2018 Mar;378:931-939.
- Wang X, Bootsma H, Terpstra J, et al. Progenitor cell niche senescence reflects pathology of the parotid salivary gland in primary Sjögren’s syndrome. Rheumatology (Oxford). 2020 Oct 1;59:3003-3013.
- Foster CS. Cicatricial pemphigoid. Trans Am Ophthalmol Soc. 1986;84:527-663.
- Fleming TE, Korman NJ. Cicatricial pemphigoid. J Am Acad Dermatol. 2000 Oct;43:571-591.
- Ahmed M, Zein G, Khawaja F, Foster CS. Ocular cicatricial pemphigoid: pathogenesis, diagnosis and treatment. Prog Retin Eye Res. 2004 Nov;23:579-592.
- Iyer G, Srinivasan B, Argarwal S. Ocular Sequelae of Stevens-Johnson Syndrome: A Comprehensive Approach. Cornea. 2020 Nov;39 Suppl 1:S3-S6.
- Lerch M, Mainetti C, Terziroli Beretta-Piccoli B, Harr T. Current Perspectives on Stevens-Johnson Syndrome and Toxic Epidermal Necrolysis. Clin Rev Allergy Immunol. 2018 Feb;54:147-176.
- Socié G, Ritz J. Current issues in chronic graft-versus-host disease. Blood. 2014 Jul 17;124:374-384.
- Dua HS, Azuara-Blanco A. Limbal stem cells of the corneal epithelium. Surv Ophthalmol. 2000 Mar-Apr;44:415-425.
- Jalbert I, Stapleton F, Papas E, Sweeney DF, Coroneo M. In vivo confocal microscopy of the human cornea. Br J Ophthalmol. 2003 Feb;87:225-236.
- Sacchetti M, Lambiase A. Diagnosis and management of neurotrophic keratitis. Clin Ophthalmol. 2014 Mar 19;8:571-579.
- Mackie IA. Neuroparalytic keratitis. In Fraunfelder F, Roy FH, Meyer SM, eds. Current Ocular Therapy. Philadelphia: WB Saunders; 1995.
- Hossain P. The corneal melting point. Eye (Lond). 2012 Aug;26:1029-1030.
- Perry HD, Golub LM. Systemic tetracyclines in the treatment of non-infected corneal ulcers: a case report and proposed new mechanism of action. Ann Ophthalmol. 1985 Dec;17:742-744.
- Hicks CR, Crawford GJ. Melting after keratoprosthesis implantation: the effects of medroxyprogesterone. Cornea. 2003 Aug;22:497-500.
- Lambiase A, Rama P, Aloe L, Bonini S. Management of neurotrophic keratopathy. Curr Opin Ophthalmol. 1999 Aug;10:270-276.
- Oyster Point Pharma Inc. Oyster Point Pharma announces FDA approval of Tyrvaya™ (Varenicline Solution) nasal spray for the treatment of the signs and symptoms of dry eye disease. [Press release] 2021 Oct 18. Available at investors.oysterpointrx.com/news-releases/news-release-details/oyster-point-pharma-announces-fda-approval-tyrvayatm-varenicline . Accessed May 30, 2022.
- Ogbru O, Davis CP. Biologics (Biologic Drug Class). MedicineNet. Available at medicinenet.com/biologics_biologic_drug_class/article.htm#what_is_a_biologic_drug_biologics . Accessed May 7, 2022.
- Geerling G, MacLennan S, Hartwig D. Autologous serum eye drops for ocular surface disorders. Br J Ophthalmology. 2004 Nov;88:1467-1474.
- Celebi AR, Ulusoy C, Mirza GE. The efficacy of autologous serum eye drops for severe dry eye syndrome: A randomized double-blink crossover study. Graefes Arch Clin Exp Ophthalmol. 2014 Apr;252:619-626.
- Jirsova K, Brejchova K, Krabcova I, et al. The application of autologous serum eye drops in severe dry eye patients; subjective and objective parameters before and after treatment. Curr Eye Res. 2014 Jan;39:21-30.
- De Pascale MR, Lanza M, Sommese L, Napoli C. Human serum eye drops in eye alterations: an insight and a critical analysis. J Ophthalmol. 2015;2015:396-410.
- Fox RI, Chan R, Michelson JB, Belmont JB, Michelson PE. Beneficial effect of artificial tears made with autologous serum in patients with keratoconjunctivitis sicca. Arthritis Rheum. 1984 Apr;27:459-461.
- Karpecki, PM. Re-think autologous serum. Rev Optom. 2018 Oct 15. Available at reviewofoptometry.com/article/rethink-autologous-serum . Accessed June 8, 2022.
- Bernabei F, Roda M, Buzzi M, Pellegrini M, Giannaccare G, Versura P. Blood-Based Treatments for Severe Dry Eye Disease: The Need of a Consensus. J Clin Med. 2019 Sep 17;8:1478.
- Ripa M, Jabbehdari S, Yazdanpanah G, et al. The Role of Multisystem Disease in Composition of Autologous Serum tears and ocular Surface symptom improvement. Ocul Surf. 2020 Jul;18:499-504.
- Harritshøj LH, Nielsen C, Ullum H, Hansen MB, Julian HO. Ready-made allogeneic ABO-specific serum eye drops: production from regular male blood donors, clinical routine, safety and efficacy. Acta Ophthalmol. 2014 Dec;92:783-786.
- Ma IH, Chen LW, Tu WH, Lu CJ, Huang CJ, Chen WL. Serum components and clinical efficacies of autologous serum eye drops in dry eye patients with active and inactive Sjogren syndrome. Taiwan J Ophthalmol. 2017 Oct-Dec;7:213-220.
- Wang M, Yennam S, McMillin J, et al. Combined therapy of ocular surface disease with plasma rich in growth factors and scleral contact lenses. Ocul Surf. 2022 Jan;23:162-168.
- Ali TK, Gibbons A, Cartes C, et al. Use of autologous serum tears for the treatment of ocular surface disease from patients with systemic autoimmune diseases. Am J Ophthalmol. 2018 May;189:65-70.
- Talmi YP, Sigler L, Inge E, Finkelstein Y, Zohar Y. Antibacterial properties of human amniotic membranes. Placenta. 1991 May-Jun;12:285-288.
- Tseng SCG. HC-HA/PTX3 purified from amniotic membrane as novel regenerative matrix: insight into relationship between inflammation and regeneration. Invest Ophthalmol Vis Sci. 2016 Apr 1;57:ORSFh1-ORSFh8.
- McDonald MB, Sheha H, Tighe S, et al. Treatment outcomes in the DRy Eye Amniotic Membrane (DREAM) study. Clin Ophthalmol. 2018 Apr 9;12:677-681.
- Cheng AM, Zhao D, Chen R, Yin HY, Tighe S, Sheha H, et al. Accelerated Restoration of Ocular Surface Health in Dry Eye Disease by Self-Retained Cryopreserved Amniotic Membrane. Ocul Surf. 2016 Jan;14:56-63.
- John T, Tighe S, Sheha H, Hamrah P, Salem ZM, Cheng AMS, et al. Corneal Nerve Regeneration after Self-Retained Cryopreserved Amniotic Membrane in Dry Eye Disease. J Ophthalmol. 2017;2017:6404918.
- Dompe. Oxervate clinical trials. Available at oxervate.com/hcp/clinical-trial-data . Accessed June 8, 2022.
- Pedrotti E, Bonacci E, Chierego C, et al. Eight months follow-up of corneal nerves and sensitivity after treatment with cenegermin for neurotrophic keratopathy. Orphanet J Rare Dis. 2022 Feb 21;17:63.
- Kwon J, Surenkhuu B, Raju I, et al. Pathological consequences of anti-citrullinated protein antibodies in tear fluid and therapeutic potential of pooled human immune globulin-eye drops in dry eye disease. Ocul Surf. 2020 Jan;18:80-97.
- Pullum KW, Whiting MA, Buckley RJ. Scleral contact lenses: the expanding role. Cornea. 2005 Apr;24:269-277.
- Shorter E, Harthan J, Nau CB et al. Scleral lenses in the management of corneal irregularity and ocular surface disease. Eye Contact Lens. 2018 Nov;44:372-378.
- Visser ES, Van der Linden BJ, Otten HM, Van der Lelij A, Visser R. Medical applications and outcomes of bitangential scleral lenses. Optom Vis Sci. 2013 Oct;90:1078-1085.
- Ciralsky JB, Chapman KO, Rosenblatt MI, et al. Treatment of refractory persistent corneal epithelial defects: a standardized approach using continuous wear PROSE therapy. Ocul Immunol Inflamm. 2015 Jun;23:219-224.
- Portnoy SL, Insler MS, Kaufman HE. Surgical management of corneal ulceration and perforation. Surv Ophthalmol. 1989 Jul-Aug;34:47-58.
- Khokhar S, Natung T, Sony P, Sharma N, Agarwal N, Vajpayee RB. Amniotic membrane transplantation in refractory neurotrophic corneal ulcers: a randomized, controlled clinical trial. Cornea. 2005 Aug;24:654-660.
- Cosar CB, Cohen EJ, Rapuano CJ, et al. Tarsorrhaphy: clinical experience from a cornea practice. Cornea. 2001 Nov;20:787-791.
- Ellis MF, Daniell M. An evaluation of the safety and efficacy of botulinum toxin type A (BOTOX) when used to produce a protective ptosis. Clin Exp Ophthalmol. 2001 Dec;29:394-399.
- Moura-Coelho N, Medel R, Cunha JP, et al. Tarsorrhaphy – Principles and Clinical Applications. J EuCornea. 2022 Mar;11. Available at eucornea.scholasticahq.com/article/33291-tarsorrhaphy-principles-and-clinical-applications . Accessed June 8, 2022.
- Zemba M, Stamate AC, Tataru CP, Branisteanu DC, Balta F. Conjunctival flap surgery in the management of ocular surface disease (Review). Exp Ther Med. 2020 Oct;20:3412-3416.
- Park JK, Charlson ES, Leyngold I, Kossler AL. Corneal Neurotization: A Review of Pathophysiology and Outcomes. Ophthalmic Plast Reconstr Surg. 2020 Sep/Oct;36:431-437.
- Catapano J, Fung SSM, Halliday W, et al. Treatment of neurotrophic keratopathy with minimally invasive corneal neurotisation : long-term clinical outcomes and evidence of corneal reinnervation. Br J Ophthalmol. 2019 Dec;103:1724-1731.




